Synthetic Nicotine Manufacturing Process and Quality Control Essentials
Synthetic Nicotine has emerged as a pivotal component in modern nicotine-based products. From vaping devices to nicotine pouches, its ultra-high purity and controlled production processes make it an invaluable alternative to natural nicotine. But what sets Synthetic Nicotine apart lies in its manufacturing precision and rigorous quality control standards. This blog will explore the essentials of Synthetic Nicotine production, focusing on the key processes, certifications, and strategies to ensure quality for manufacturers, suppliers, and wholesalers.
Route-A Versus Route-B Chemical Synthesis and Their Impurity Profiles
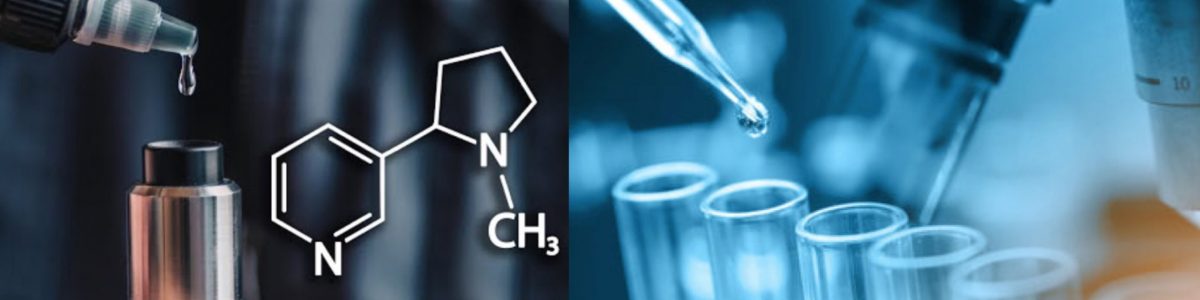
The chemical synthesis of Synthetic Nicotine primarily follows two distinct routes, referred to as Route-A and Route-B. These pathways differ in the reagents and catalysts used, directly impacting the resulting impurity profiles. Route-A typically employs a racemic approach, resulting in a mixture of S- and R-nicotine stereoisomers, which then undergo separation to enhance purity. On the other hand, Route-B utilizes enantioselective catalysis to produce a high S-isomer ratio, significantly reducing subsequent purification steps. Synthetic Nicotine manufacturers favor Route-B for its efficiency and reduced chemical waste. However, both routes require robust impurity testing to maintain USP/EP compliance, ensuring a purity level of ≥ 99.9%.
In-Process Analytics: Chiral Purity Testing for Synthetic Nicotine Suppliers
One of the hallmarks of high-quality Synthetic Nicotine lies in its chiral purity. The S-nicotine isomer is primarily responsible for the therapeutic and sensory effects sought by manufacturers. Chiral purity testing during production is crucial to ensure that the final product matches the desired specifications. Techniques like high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS) are often employed to analyze stereoisomer ratios accurately. Synthetic Nicotine suppliers leverage these methods to maintain their commitment to delivering pharmaceutical-grade nicotine. Regular testing and in-process analytics ensure not only compliance with international safety standards but also consistency in each production batch.
GMP and ISO-Certification Checkpoints for Synthetic Nicotine Wholesale Production Lines
For Synthetic Nicotine wholesale operations, adherence to Good Manufacturing Practices (GMP) and ISO certifications is non-negotiable. These standards guarantee that the production process minimizes contamination risks, employs validated methodologies, and maintains detailed documentation for traceability. Key checkpoints include the validation of raw materials, periodic equipment calibration, and comprehensive employee training. Additionally, independent audits are often conducted to verify compliance and highlight areas for improvement. Synthetic Nicotine manufacturers with ISO 9001 and GMP certifications often have a competitive edge in the global marketplace, as these certifications signify reliability and commitment to quality excellence.
We specialize in manufacturing Synthetic Nicotine high-purity 99.90% ( CAS No.: 54-11-5 ) with the highest technology and stable quality control , our factory has sophisticated technology production route to produce this product ,welcome to inquiry it . Please contact us by e-mail sales@wutaichem.com for quotation requests.
http://www.wutaichem.com/synthetic-nicotine-manufacturing-process-and-quality-control-essentials/Synthetic Nicotine is used for E-Cigarettes