Is synthetic nicotine regulated by the FDA?
The short answer is now yes. On March 11, 2022, Congress passed, and the President signed language into law that clarified that Synthetic Nicotine products must be regulated by the FDA’s Center for Tobacco Products in the same way that tobacco-derived nicotine products are regulated.
When these products first came onto the market, some companies claimed that Synthetic Nicotine products did not have to be regulated by the FDA. While this was not accurate, the FDA was slow to determine how it would regulate these products and it was unclear whether these products would be regulated as tobacco products or drugs.
Products containing tobacco or tobacco-derived nicotine must be regulated by the Center for Tobacco Products which is guided by the public health standard. Because Synthetic Nicotine is not derived from tobacco, it was a question as to whether they would have been regulated by the Center for Drug Evaluation and Research which uses a different standard based on safety and effectiveness.
With the change in law, it closes the regulatory gap surrounding Synthetic Nicotine products and ensures that Synthetic Nicotine and tobacco-derived nicotine products are all regulated under the Center for Tobacco Products using the same standard. (Note: Some products that are considered drugs, such as Nicotine Replacement Therapies used to help people quit tobacco, are regulated under FDA’s Center for Drug Evaluation and Research and must go through CDER’s approval process for safety and effectiveness.)
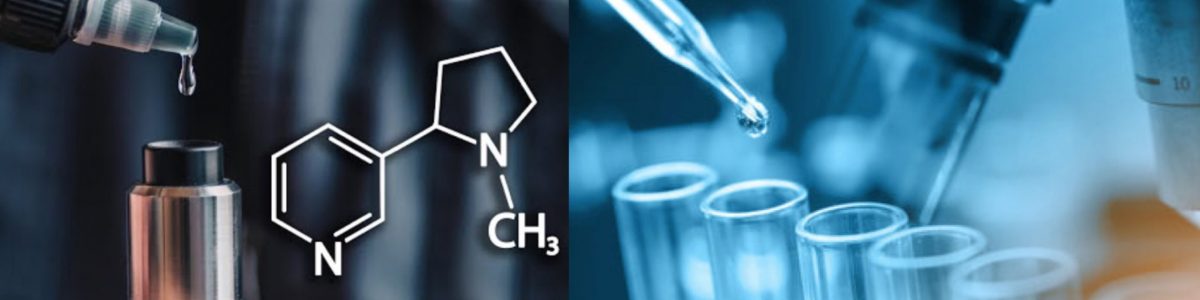
We specialize in manufacturing Synthetic Nicotine high-purity 99.90% ( CAS No.: 54-11-5 ) with the highest technology and stable quality control , our factory has sophisticated technology production route to produce this product ,welcome to inquiry it . Please contact us by e-mail sales@wutaichem.com for quotation requests.
http://www.wutaichem.com/is-synthetic-nicotine-regulated-by-the-fda/Synthetic Nicotine is used for Quitting Smoking